White blood cells push their way through barriers to get to infection sites
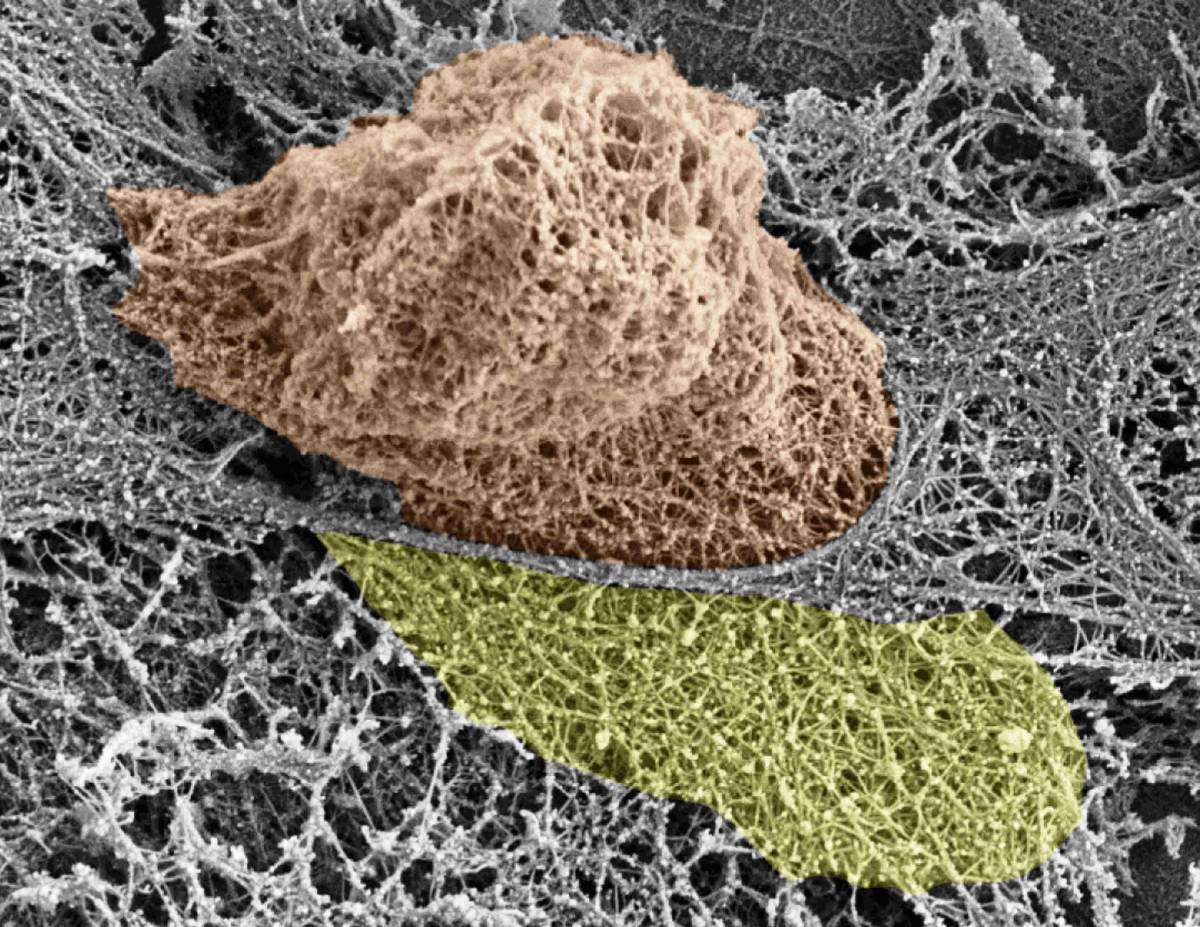
One of the mysteries of the living body is the movement of cells – not just in the blood, but through cellular and other barriers. Such movement takes place continually, in both healthy activity and disease. New research in the Weizmann Institute of Science has shed light on the subject, especially on the movement of immune cells that race to the sites of infection and inflammation. The study revealed that these cells – white blood cells – actively open large gaps in the internal lining of the blood vessels, so they can exit through the vessel walls and rapidly get to areas of infection.
Prof. Ronen Alon and his group in the Weizmann Institute’s Immunology Department discovered how various white blood cells push their way through the lining of the blood vessels when they reach their particular “exit ramps.” Using their nuclei to exert force, they insert themselves between – as well as into – the cells in the vessel walls called endothelial cells. Dismantling specific filaments within the cytoskeletons – the internal skeletons – of the endothelial cells is what creates the large holes, which are several microns in diameter.
The nucleus is better known as the repository of the cells’ DNA but, as Alon explains, it is also the largest, most rigid structure in the cell. Although it tends to be relatively soft and flexible in white blood cells, the nucleus, when driven by motors specifically engaged for this function, is tough enough to push through the barrier imposed by the blood vessel walls.
To get a complete picture of this crucial activity, the scientists tracked the internal skeletons of endothelial cells as they were crossed by immune cells in real time. They followed the behavior of the nuclei of various white blood cells during active squeezing, as well as the fate of the various types of actin fibers that make up the endothelial cell skeletons. The researchers used a number of methods, including fluorescence and electron microscopy, in collaboration with Dr. Eugenia Klein of the Microscopy Unit; a unique system in Alon’s lab for simulating blood vessels in a test tube; and in vivo imaging with Prof. Sussan Nourshargh of Queen Mary University of London. The results of this research, conducted in Alon’s lab by research students Sagi Barzilai and Francesco Roncato and postdoctoral fellow Dr. Sandeep Kumar Yadav, were recently reported in Cell Reports.
Dismantling specific filaments within the internal skeletons of the endothelial cells creates large holes, several microns in diameter
The interactions between white blood cells and the endothelial cells lining the blood vessels near sites of infection and inflammation have been widely studied. But the new research revealed an unexpectedly active role for immune cells – one that had previously been assigned to the endothelial cells: the ability to breach blood vessel walls. Indeed, common wisdom in this field had held that the endothelial cells must help immune cells squeeze through by contracting themselves like small muscles. The present study, however, found no evidence for such contraction-based help. “Immune cells play a critical role in getting their own bodies across the barrier presented by blood vessel walls,” says Alon. “Our study shows that the endothelial cells, which were thought to be dynamic assistants in this process of crossing of blood vessel walls, are really more responders to the ‘physical work’ invested by the white blood cell motors and nuclei in generating gaps and crossing through blood vessels.”
Significance for cancer research
For over two decades Alon and his lab group have been investigating the movement of immune cells along and through inflamed blood vessels. Among other things, they discovered the “stop” signs – biochemical markers – that help these cells navigate to specific sites of infection, and the tiny “legs” that white blood cells use to help them hold on to the endothelial cells lining the blood vessels and search for additional exit signs.
In addition to increasing the basic understanding of how the various arms of the immune system reach their sites of differentiation and activity, these findings may aid in cancer research. Malignant cells that travel to distant sites in the body through the bloodstream may employ some of guidance biochemicals and mechanisms used by the white blood cells to pass out of the blood vessels. “We believe that small subsets of metastatic tumor cells have the ability to adopt the mechanisms used by immune cells to exit the blood vessels into the lungs, the bone marrow, the brain and other organs. If this is true, we might be able to identify these subsets and target them before these cells leave their original tumor sites and invade distant organs,” says Alon.

Recent Comments